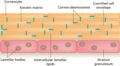
Figure 13.1
A schematic representation of the stratum corneum as a brick wall. The corneocytes act as the bricks, with the intercellular lipids providing the ceme...
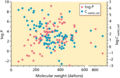
Figure 13.5
Water solubilities and log P values of 91 drugs approved for topical and transdermal administration in Europe and/or the USA as a function of their mo...
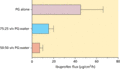
Figure 13.9
Flux of ibuprofen into human stratum corneum in vivo from three formulations [ ] in which the drug was present at its saturation concentration. PG, ...
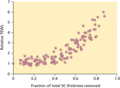
Figure 13.2
Transepidermal water loss (TEWL) across human skin in vivo as the stratum corneum (SC) is progressively removed by adhesive tape stripping [ ]. The ...

Figure 13.6
Estimated maximum flux ( J max ) values for approved topical and transdermal drugs as functions of: (a) their size (MW); (b) their lipophilicity (log ...

Figure 13.10
Stimulated Raman spectroscopic images of (a) solid ibuprofen and (b) betamethasone valerate particles crystallized on and within the outer stratum cor...
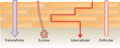
Figure 13.3
Potential penetration pathways across the stratum corneum [ ].

Figure 13.7
Methods, both accepted and under consideration by the regulatory authorities (see text for details), for the determination of topical bioavailability ...
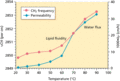
Figure 13.4
Correlation between the permeability coefficient (k p ) of water across the stratum corneum (SC) with the conformational order of the SC intercellular...
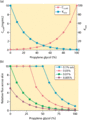
Figure 13.8
(a) Formulation of a lipophilic drug (e.g. a corticosteroid) of low water solubility, in an aqueous gel [ ]. The profiles illustrate the manner in whi...

